A Treatment Option for Infantile Hemangioma Is Here
- 60% of infantile hemangioma completely or nearly completely resolved by 6 months vs 4% for placebo
- 88% of infantile hemangioma showed improvement after 5 weeks of treatment.*
*Based on independent-investigator assessment of telangiectasis, color, textural change and distortion of anatomical landmarks or skin contours compared to baseline.
The Discovery of Propranolol in the Treatment of Infantile Hemangioma
In 2007, while treating a baby for Infantile Hemangioma with oral steroids, the baby presented a cardiomyopathy (a complication of this treatment). Therefore, the child was treated with propranolol. Dr. Christine Léauté-Labréze and her colleagues discovered that propranolol hydrochloride (the active ingredient in HEMANGEOL) could also control the growth of hemangiomas. It is believed that the drug may have an effect on proliferating infantile hemangiomas through vasoconstriction, inhibition of angiogenesis, and induction of apoptosis.1
This discovery prompted a randomized, controlled clinical trial to investigate the safety and efficacy of propranolol hydrochloride (HEMANGEOL) for infantile hemangioma, with the goal of developing a pediatric formulation and strength that met regulatory guidelines and requirements, including the European Medicines Agency (EMA) and U.S. Food and Drug Administration (FDA).2,3
1. Fette A. Propranolol in use for treatment of complex infant hemangiomas: literature review regarding current guidelines for preassessment and standards of care before initiation of therapy. Scientific World Journal. 2013:1-9.
2. Drolet BA, Frommelt PC, Chamlin SL, et al. Initiation and use of propranolol for infantile hemangioma: report of a consensus conference. Pediatrics. 2013;131(1):128-140.
3. European Medicines Agency website. European Medicines Agency decision. https://www.ema.europa.eu/docs/en_GB/document_library/PIP_decision/WC500141483.pdf. Accessed January 21, 2023
Proven Efficacy
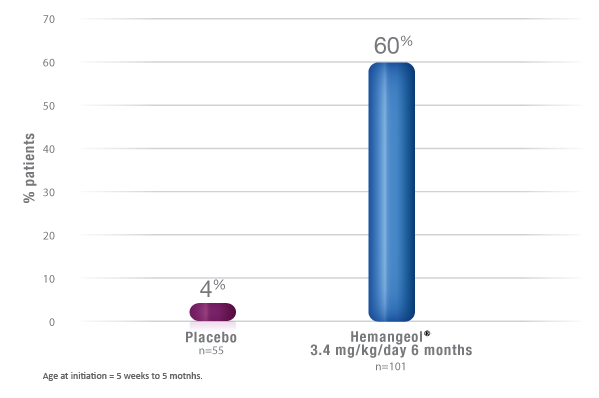
Complete or Nearly Complete Resolution After 6 Months of Therapy
Statistically significant response was shown in patients (5 weeks to 5 months of age with proliferating infantile hemangiomas) treated with HEMANGEOL versus placebo.
In a pivotal Phase II/III clinical trial (n=460), 4 regimens of propranolol hydrochloride were compared: 1.2 or 3.4 mg/ kg/day in twice daily divided doses for 3 or for 6 months as compared to placebo.
Among patients treated with HEMANGEOL 3.4 mg/kg/day for 6 months
- 60% of infants receiving HEMANGEOL vs. 4% of those in the placebo group
met the primary endpoint of complete or nearly complete resolution of infantile hemangioma (P<0.0001) - 10% of successful patients at week 24 required retreatment of their infantile hemangioma
- Life-threatening infantile hemangioma, function-threatening infantile hemangioma, and ulcerated infantile hemangioma with pain and lack of response to simple wound care measures were excluded
- 88% of patients on HEMANGEOL showed improvement of infantile hemangioma after 5 weeks of treatment*
**Based on independent-investigator assessment of telangiectasis, color, textural change and distortion of anatomical landmarks or skin contours compared to baseline.
In a second open-label clinical study (n=23), target lesions resolved by 3 months in 36% of infants with proliferating infantile hemangioma, including:
- Function-threatening infantile hemangioma
- Infantile hemangioma in certain anatomic locations that often leave scars or deformity
- Large facial infantile hemangioma
- Smaller infantile hemangioma in exposed areas
- Severe ulcerated infantile hemangioma
- Pedunculated infantile hemangioma
Efficacy At a Glance
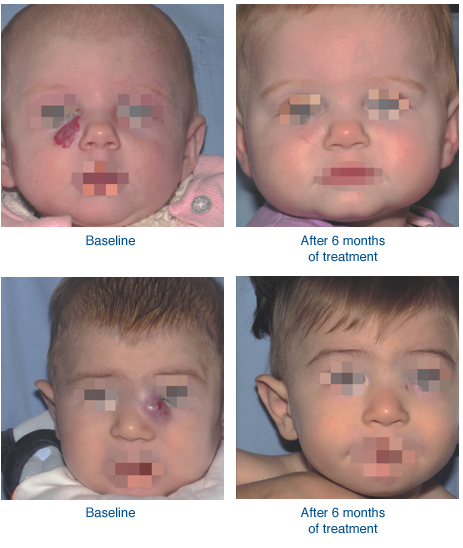
Before and After HEMANGEOL Treatment
*individual results may vary
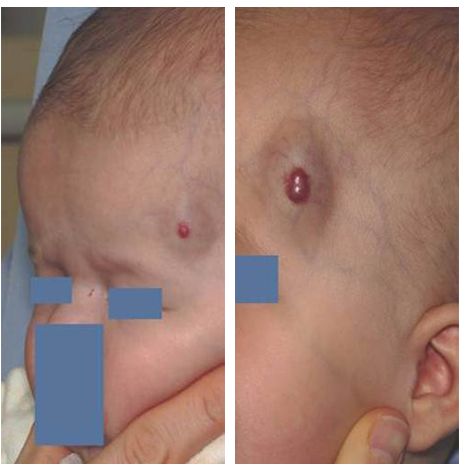
Before and After HEMANGEOL Treatment
*individual results may vary
Safety Profile
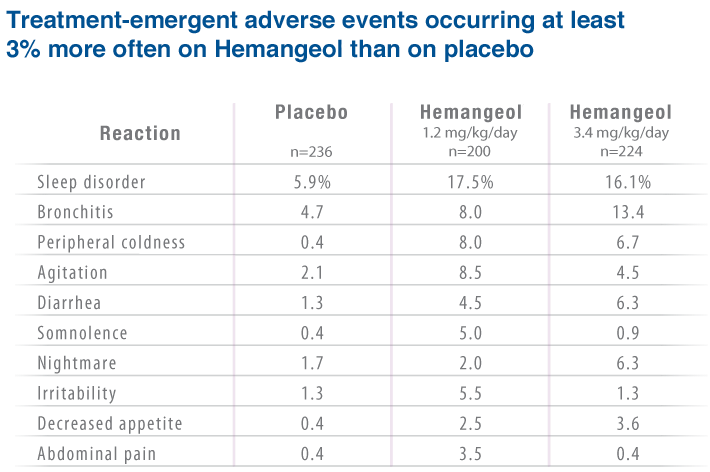
The most frequently reported adverse reactions (≥10%) in infants treated with HEMANGEOL were:
- Sleep disorders
- Aggravated respiratory tract infections such as bronchitis and bronchiolitis associated with cough and fever
- Diarrhea
- Vomiting
The following adverse events were observed with an incidence of less than 1%:
- Cardiac disorders
- Decreased blood glucose
- Decreased heart rate
- Urticaria
- Alopecia
Fewer than 2% of treated patients discontinued treatment due to adverse reactions
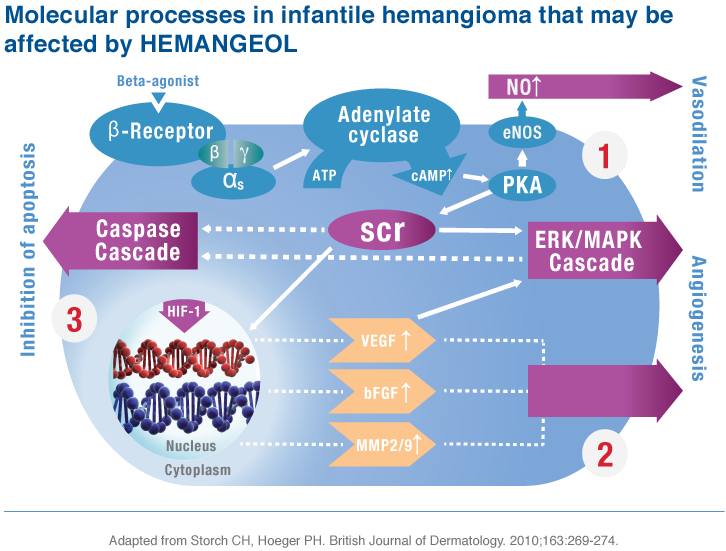
Mechanism of Action Hypothesis
The mechanism of action of HEMANGEOL (propranolol hydrochloride) in infantile hemangioma is not well understood.
The hypothesis is that the drug’s effect on proliferating infantile hemangioma can be attributed to 3 molecular mechanisms, leading to1:
- A local hemodynamic effect (reduction in blood flow)
- An anti-angiogenic effect (reduction of growth factors)
- An apoptosis triggering effect on capillary endothelial cells (increase rate of infantile hemangioma cell death)